Researchers at Vanderbilt University Medical Center are leading an international clinical trial to examine the functional benefits of a combined therapy to treat acute ischemic stroke, a type of stroke where a blood clot becomes lodged in an artery within the brain, reducing blood flow and depriving brain cells of oxygen. Eighty-seven percent of strokes are ischemic.

The THERAPY trial involves use of the standard-of-care, clot-busting intravenous drug tPA (tissue plasminogen activator) and an interventional device treatment, the Penumbra System, that gently suctions away blood clots to restore blood flow to the brain’s affected area.
The trial will evaluate a combined therapy using IV tPA and interventional clot removal with the Penumbra System, compared with IV tPA treatment alone.
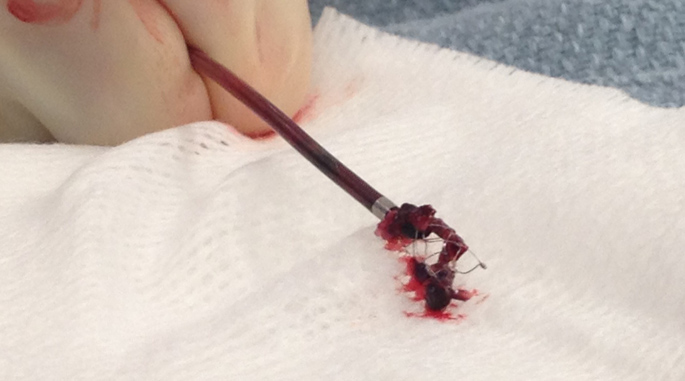
During the minimally invasive procedure a catheter is threaded to the clot and the Penumbra System’s technology gently suctions away the mass to restore blood flow. The Penumbra System, which was FDA-approved in 2007, has been used in approximately 15,000 stroke treatment procedures in the United States.
Principal investigator J Mocco, M.D., associate professor of Neurological Surgery, said plans call for the trial to involve 75 stroke treatment centers worldwide, with a target enrollment of 692 patients.

“When patients come in with a stroke they are distressed,” Mocco said. “They can’t move half their body, they often can’t talk, and they can’t understand language. They are often headed toward permanent disability, or death.”
“The device evaluated in this trial works like a straw, it literally sucks the clot out. In our recent experience at Vanderbilt, almost half of the treated patients are completely independent afterwards. These are patients who otherwise would likely be devastated with severe disability.”
Stroke is the fourth leading cause of death in the U.S. and the leading cause of long-term adult disability, affecting 800,000 Americans every year. Common stroke symptoms include numbness, weakness or paralysis along one side of the body, slurred speech, and difficulty understanding speech and/or trouble walking.
The standard of care for acute ischemic stroke in most stroke centers involves administration of IV tPA within 3 to 4.5 hours of the onset of symptoms. However, most patients do not arrive in time, and the treatment may fail if a clot is too large or difficult to dissolve. If this occurs, alternative minimally invasive, inside-the-artery clot removal can be used up to eight hours after onset of symptoms.
The trial will also examine the use of certain patient selection criteria to determine if interventional therapy is appropriate. Recent advancements in CT scan imaging have helped physicians better assess the size and location of a patient’s clot and whether inside-the-artery clot removal will work.
“If successful, the THERAPY trial will help to identify a patient population most likely to benefit from interventional therapy, as well as reveal a treatment paradigm that may have a true impact on improving the standard of care for ischemic stroke patients,” Mocco said.
“Acute stroke intervention technology has grown by leaps and bounds. In fact, it has only been in the past decade that any technology has been available to mechanically remove clots causing stroke,” Mocco said.