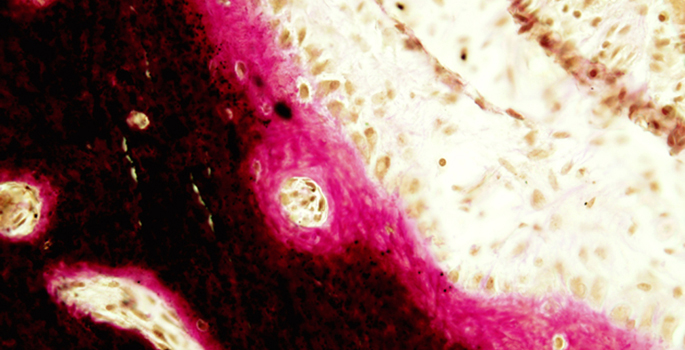
The lengthening of the long bones – arms, legs and ribs – during development, and their healing following fracture, occurs in a process called endochondral bone formation. Fibroblast growth factor receptor (FGFR) signaling is important for proper bone elongation and healing, but the machinery that fine-tunes FGFR signaling in chondrocytes – the cartilage-forming cells that mature into calcified bone – is incompletely defined.
Florent Elefteriou, Ph.D., graduate student Matthew Karolak and colleagues at the Vanderbilt Center for Bone Biology explored the role of neurofibromin in FGFR signaling in maturing chondrocytes in vivo. They report May 1 in Human Molecular Genetics that neurofibromin acts as a brake on FGFR signaling in chondrocytes. They provide evidence that neurofibromin in mature chondrocytes is required for normal extension and organization of proliferative columns in the growth plate, and for its degradation by osteoclasts, the bone-resorbing cells.
The findings have relevance to genetic diseases affecting embryonic development of the skeleton and to conditions impacting bone healing in adults, such as neurofibromatosis type 1.
This research was supported by the National Institutes of Health (GM007628, AR055966), the Children’s Tumor Foundation and the Department of Defense.
Send suggestions for articles to highlight in Aliquots and any other feedback about the column to aliquots@vanderbilt.edu