Researchers at Vanderbilt University and the University of Michigan have discovered a critical factor in the rheostatic control of the body weight by the brain.
Their findings, reported this week in the journal Science Advances, help explain the weight gain that occurs during pregnancy and could lead to a new approach for treating obesity.
Normally, brain signals to regulate the balance of energy intake and expenditure aim to keep body weight stable near a value called the body weight “set point.” In response to metabolic challenges such as starvation, an energy-rich diet, pregnancy or menopause, the brain ought to reset to a new appropriate energy balance/body weight set point.
Important signaling pathways, including leptin and the melanocortin-4 receptor (MC4R), have been shown to be involved in this balance. Defects in either pathway result in increased fat storage and body weight.
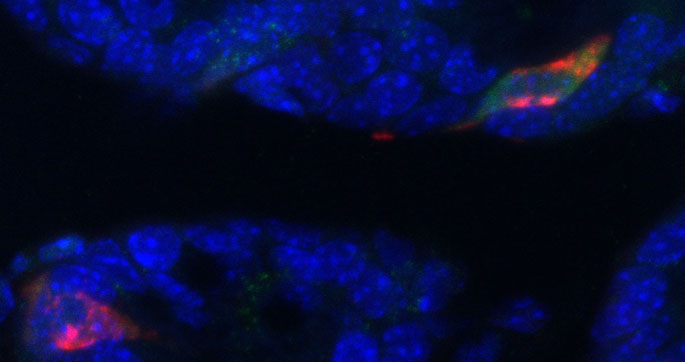
The movement from one energy set point to another, called energy rheostasis, is regulated through mechanisms that have eluded scientists for decades. The study by Vanderbilt and Michigan scientists showed that a previously ill-defined brain protein, MC3R, plays a role in energy rheostasis.

Under normal conditions this protein can be deleted from mouse brain with minimal effects on energy balance. However, when subjected to fasting, mice without MC3R lose more weight than control counterparts. When subjected to a high-fat diet, they gain more weight.
This suggests that without MC3R’s bidirectional control of the energy “rheostat,” the body responds abnormally to caloric restriction or a calorie-rich diet, as well as to pregnancy. MC3R thus ensures the balance of energy and fat in the body does not drift too far in either direction when the body experiences some sort of metabolic stress.
“This role in rheostasis makes MC3R a promising new drug target for treating obesity,” said Masoud Ghamari-Langroudi, PhD, research assistant professor of Molecular Physiology and Biophysics at Vanderbilt and the article’s lead author.
“When we eat less and exercise more to lose weight, our bodies sense when the energy balance has tipped below the set lower boundary and try to adjust by using less energy or and increasing appetite,” he said. “This lower boundary is what makes it difficult to keep weight off.”
By recording brain cell activity from the mice, Ghamari-Langroudi observed a reciprocal inhibitory interaction between MC3R and MC4R. Activation of MC3R resulted in inhibition of MC4R through the release of the inhibitory neurotransmitter GABA.
“This finding deepens our understanding of how energy balance is regulated,” the paper’s senior author, Roger Cone, PhD, director of the University of Michigan Life Sciences Institute, said in a news release.
“A drug that targets MC3R has the potential to work as a diet aid by reducing the rigidity of that lower boundary,” he said. “It could enable people to keep the weight off when they improve eating and exercise habits.”