Researchers at Vanderbilt University Medical Center, Purdue University and the University of Wisconsin-Madison have isolated human monoclonal antibodies that potentially can prevent a rare but devastating polio-like illness in children linked to a respiratory viral infection.
The illness, called acute flaccid myelitis (AFM), causes sudden weakness in the arms and legs following a fever or respiratory illness. More than 600 cases have been identified since the U.S. Centers for Disease Control and Prevention began tracking the disease in 2014.
There is no specific treatment for AFM, which tends to strike in the late summer or early fall and which has been associated with some deaths. However, the disease has recently been linked to a group of respiratory viruses called enterovirus D68 (EV-D68).
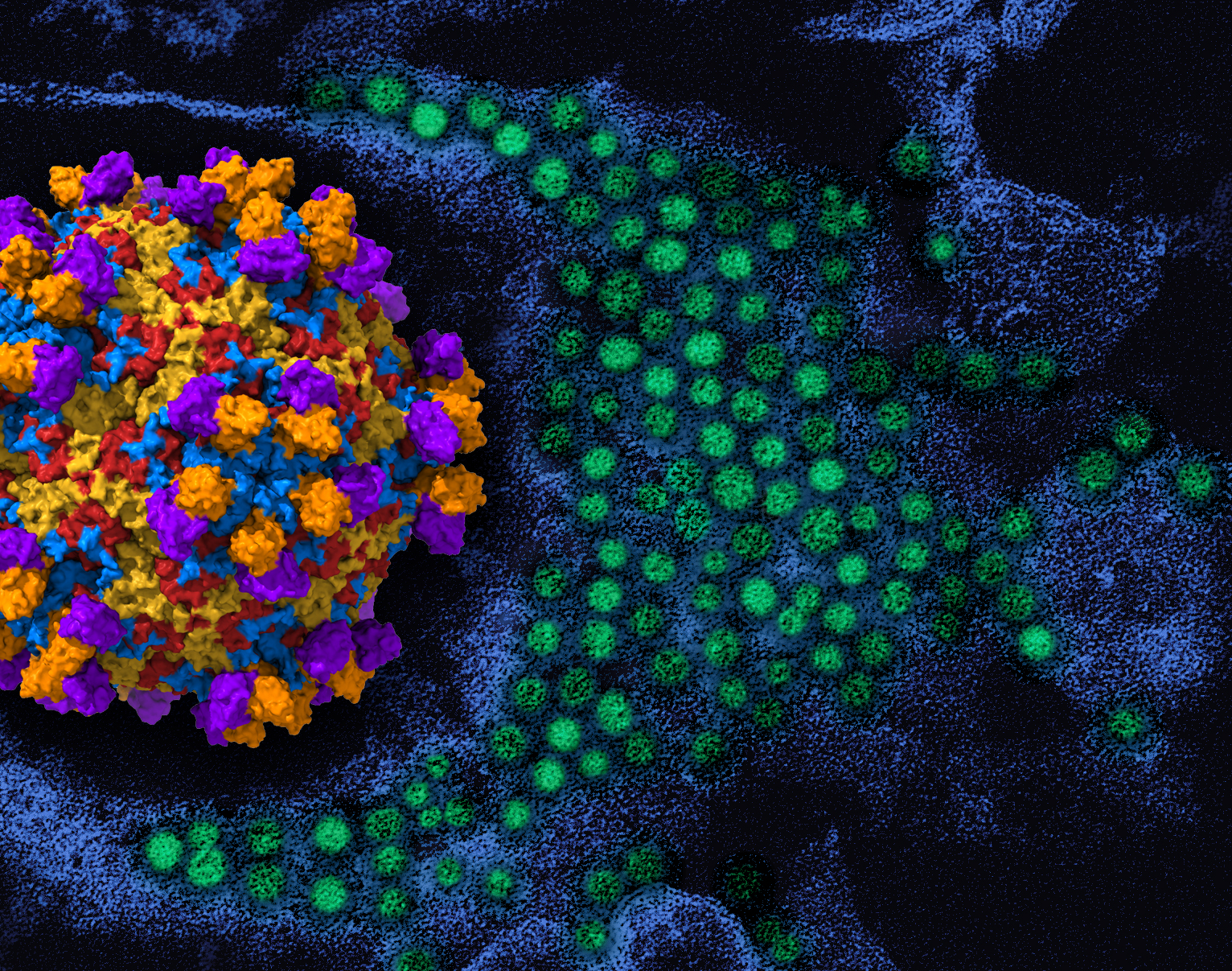
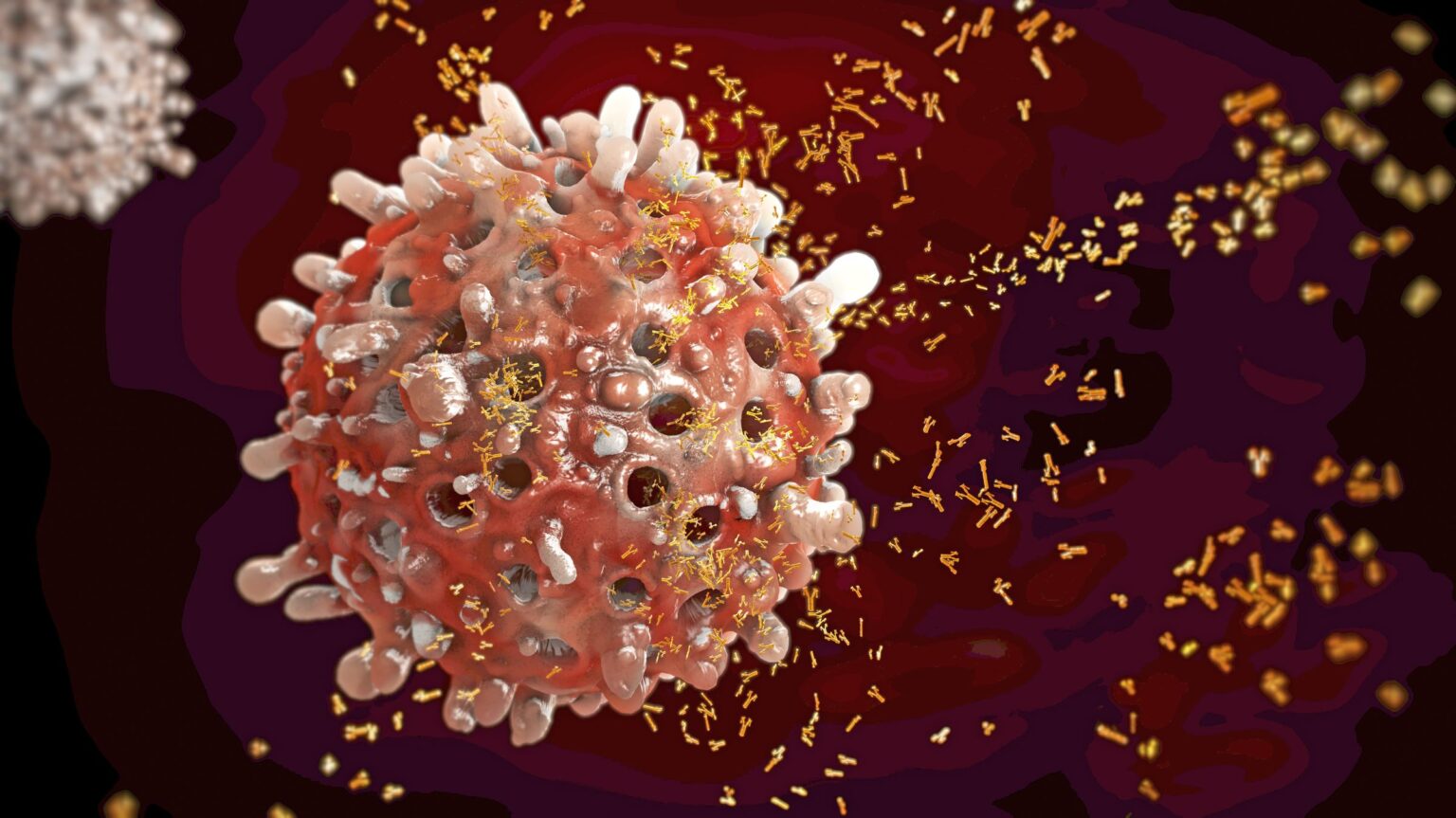
Researchers at the Vanderbilt Vaccine Center isolated antibody-producing blood cells from the blood of children who had previously been infected by EV-D68. By fusing the blood cells to fast-growing myeloma cells, the researchers were able to generate a panel of monoclonal antibodies that potently neutralized the virus in laboratory studies.
Colleagues at Purdue determined the structure of the antibodies, which shed light on how they specifically recognize and bind to EV-D68. One of the antibodies protected mice from respiratory and neurologic disease when given either before or after infection by the enterovirus.
The findings, published July 3 in the journal Science Immunology, suggest that these antibodies potentially could prevent EV-D68 infection and AFM in humans.
“We were excited to isolate potent human antibodies that inhibit this devastating polio-like virus, and these studies will form the basis for taking them forward to clinical trials,” said James Crowe, MD, director of the Vanderbilt Vaccine Center, Ann Scott Carell Professor of Pediatrics and Pathology, Microbiology and Immunology.
“Studying infectious disease from a very basic level and applying the results in an animal model of disease is very powerful; hopefully, our studies will translate to a future therapeutic for this disease in children,” said Richard Kuhn, PhD, Purdue’s Trent and Judith Anderson Distinguished Professor in Science; Krenicki Family Director, Purdue Institute of Inflammation, Immunology and Infectious Disease.
Matthew Vogt, MD, PhD, a fellow in Pediatric Infectious Diseases at VUMC and recipient of a Pediatric Infectious Diseases Society (PIDS)-St. Jude Children’s Research Hospital Fellowship, is the paper’s first author.
Other VUMC co-authors are Nurgun Kose, Lauren Williamson, Robin Bombardi, MS, Ian Setliff and Ivelin Georgiev, PhD.
National Institutes of Health grants HL069765, AI117905, HL070831, AI104317 and AI011219 and the Center for Structural Genomics of Infectious Diseases supported the study.