Using a four-dimensional microscope that allows them to watch a tissue putting itself together, researchers at Vanderbilt University Medical Center have achieved a rare feat in science — they have shattered a long-standing dogma about how the lung develops.

Their tour de force raises the possibility that one day scientists will be able to repair damaged lungs by triggering the growth of normal tissue, and that doctors will be able to prevent the development of lung disease in babies born prematurely.
“We weren’t trying to find this,” said the lead researcher, Jennifer Sucre, MD. “We were trying to create a platform to understand alveologenesis,” how the alveoli, the tiny breathing sacs in the lungs that enable the exchange of oxygen and carbon dioxide, develop.
“What we found was something completely different.”
To continue their work and to determine the molecular and cellular basis for a lung disease of prematurity called bronchopulmonary dysplasia (BPD), Sucre and her colleagues have been awarded a five-year, $3.5-million grant (HL168556) by the National Heart, Lung, and Blood Institute of the National Institutes of Health.
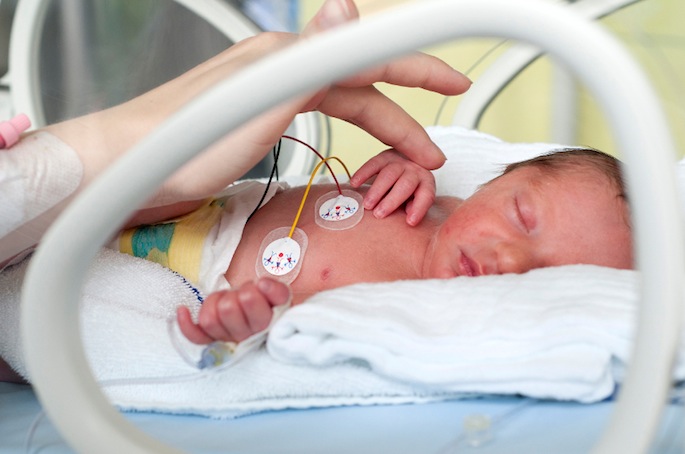
About 50% of infants born at a gestational age of 22 to 32 weeks — two to four months too early — develop BPD, a life-threatening chronic lung disease that requires respiratory support and oxygen to help them breathe.
BPD results from abnormal lung development in the setting of premature birth. Treatment of neonatal respiratory distress is a two-edged sword, however, as the oxygen and mechanical ventilation required for survival in the early days after birth can also injure delicate lung tissue.
While many premature babies can be weaned off oxygen therapy after a few days, later in life survivors of premature birth are at increased risk for developing serious breathing problems, including chronic obstructive pulmonary disease.
Given that treating infants with BPD already costs insurers and families an estimated $2.4 billion a year in the United States, “the respiratory consequences for aging survivors of prematurity present a looming public health crisis,” said Sucre, the grant’s principal investigator.
An assistant professor of Pediatrics and of Cell and Developmental Biology, Sucre’s research focus has been driven by her clinical work as a neonatologist, caring for critically ill infants in Vanderbilt’s neonatal intensive care unit (NICU).
“While my own work is inspired by preterm infants, it really applies to lung injury across the lifespan,” she said. “In this post-COVID world, we realize that all of us are vulnerable to lung injury. Thinking about how to grow new lungs is not just for babies.”
Since joining the Vanderbilt faculty in 2016, Sucre and her colleagues have developed techniques that enable them to track the complex interplay of cells involved in forming the alveoli.
In 2021 they used a technique called single-cell RNA-sequencing to construct a single-cell “atlas” of lung development in the mouse to understand the role and behavior of multiple cell types over time.
Among other findings reported by postdoctoral fellow Nicholas Negretti, PhD, as first author, the team described a previously unrecognized role of alveolar type 1 (AT1) pneumocyte cells in establishing the extracellular matrix scaffold of the developing alveolus.

Now, in partnership with Bryan Millis, PhD, research associate professor of Biomedical Engineering at Vanderbilt, they are using a 4D microscopy technique called scanned oblique plane imaging (SOPi), to create 3D video images of precision-cut slices of developing mouse lung grown in the laboratory over time — the fourth dimension.
This is where the revolutionary part comes in.
According to the traditional, 2D view of alveologenesis, septa (dividers) emerge from a layer of epithelial, endothelial, and mesenchymal cells to create and subdivide airspaces into alveoli filled with tiny blood vessels where the actual gas exchange takes place.
When slices of living neonatal mouse lung tissue are imaged by SOPi over three days, however, a totally different view emerges, one of a ballooning outgrowth of epithelial cells supported by a ring of myofibroblasts, cells that promote tissue formation.
Cube-shaped alveolar type 2 (AT2) pneumocyte cells extrude through the rings and, over the next several hours, differentiate into flattened-out AT1 cells, which are key to building a scaffold for the developing alveolus and eventually form its inner lining.
Fluorescent labels were used to distinguish between different cell types. This enabled the researchers to track cells over time, and Philip Crooke, PhD, research professor of Mathematics at Vanderbilt, to create a computational model of alveolar formation.
“The process of postnatal alveologenesis is not something we have seen before in real time,” Sucre said. “The breakthrough is that with long-term live imaging, we can watch alveologenesis in real time, and we can measure it.
“Our observations that inform this new model of lung organogenesis are changing the paradigm of questions we can ask about this fundamental process,” she added, “with implications beyond the NICU to understanding how to regenerate injured lung across the lifespan.”
Normal lung development depends on a precisely timed and coordinated set of molecular cues delivered by myofibroblasts via the Wnt signaling pathway, and by the BMP family of growth factors.
The team’s working hypothesis is that Wnt drives the extrusion of AT2 cells through the myofibroblast ring, while BMP promotes their differentiation into AT1 cells. Injury caused by inflammation or overexposure to oxygen disrupts the signals, interrupts construction of the alveolar scaffolding, and results in impaired lung function — BPD.
With the support of the NIH grant, the researchers plan to further define the roles of Wnt and BMP signaling in alveolar development and in neonatal lung injury, and test new approaches for protecting and restoring normal lung growth in babies born prematurely.
For example, knowing that overactivation of the Wnt pathway has been associated in premature infants with BPD, the researchers will test whether small-molecule inhibitors of Wnt signaling can repair damaged tissue in lung slices grown in the laboratory.
“In the future, it would be fantastic to screen the drugs we use in the NICU and test them on this system,” Sucre said.
If one drug has a side effect that disrupts alveolar development, while another drug in the same class promotes it, “we can make an informed choice with regard to the lungs, with a goal of leveraging this platform to discover new targeted therapies,” she said.
Sucre was quick to add that it has taken “a village” to push the research forward.
Other key collaborators include lab manager and research assistant Chris Jetter; Timothy Blackwell, MD, the Rudy W. Jacobson Chair in Pulmonary Medicine, professor of Medicine and Cell and Developmental Biology, and director of the Center for Lung Research; Christopher Wright, DPhil, the Louise B. McGavock Chair, professor of Cell and Developmental Biology, and director of the Vanderbilt Program in Developmental Biology; Erin Plosa, MD, assistant professor of Pediatrics; Emily Hodges, PhD, assistant professor of Biochemistry; and former Vanderbilt faculty member Brigid Hogan, PhD, professor of Cell Biology and Pediatrics, emeritus, at Duke University.
Beyond the science, Sucre marvels at the extraordinary beauty of unfolding lung cells that resemble exquisite blossoms when viewed through the 4D microscope. It is as if she is seeing the lung for the first time.
At the end of her lectures, Sucre often recites a passage from early 20th century French novelist Marcel Proust’s seminal work, “In Search of Lost Time.” “The real voyage of discovery,” Proust wrote, “consists not in seeking new landscapes, but in having new eyes.”
“The best discoveries,” she added, “are when we are surprised.”