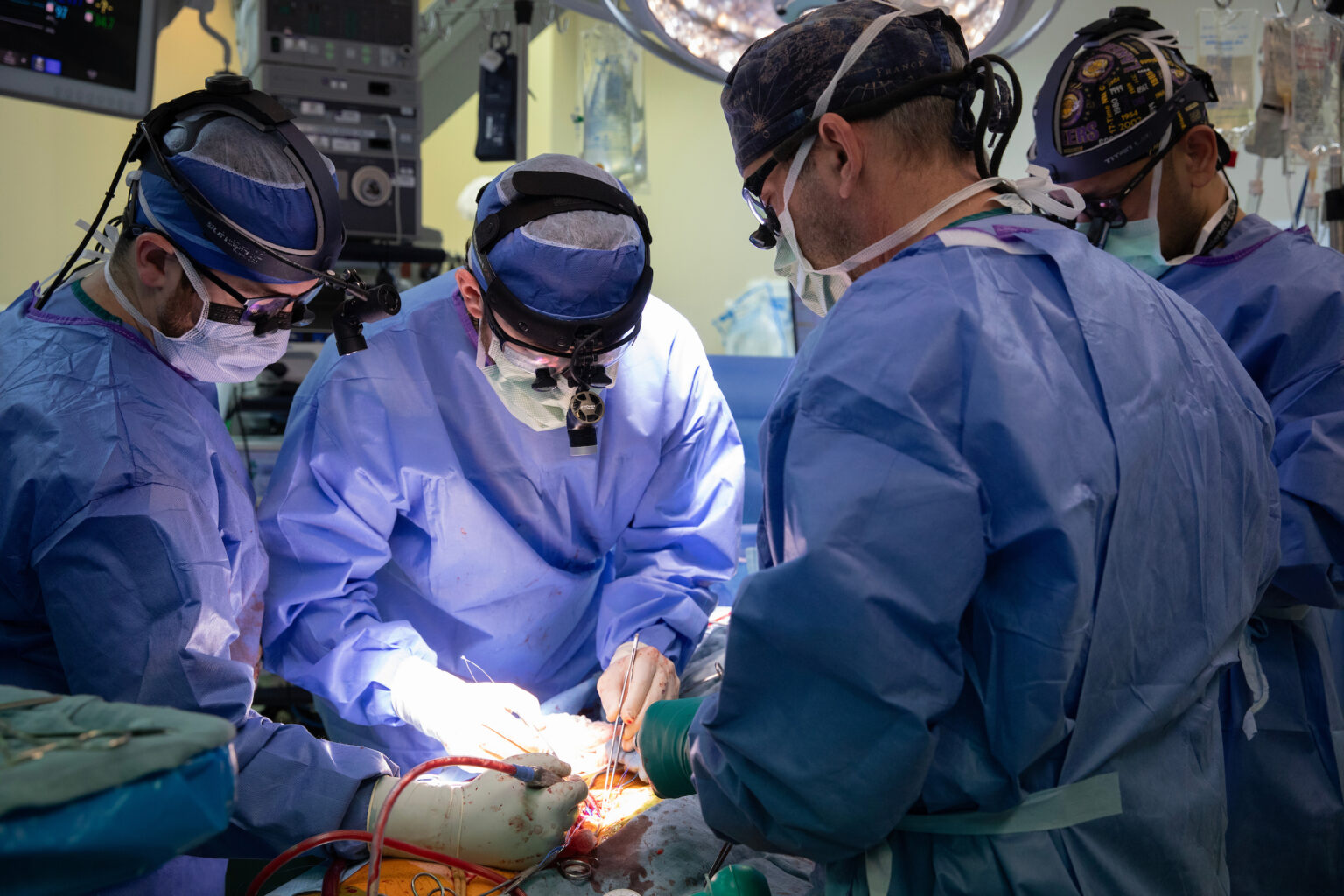
In the first such procedure, Vanderbilt University Medical Center has transplanted a DCD (donation after cardiac death) heart using XVIVO Heart Assist Transport (XHAT), a novel cold organ perfusion device. The transplant was completed in December 2023 under the direction of Ashish Shah, MD, professor and chair of the Department of Cardiac Surgery, as part of a multi-institutional clinical trial.
Over the last four years, Vanderbilt’s heart transplant program has been a leader in utilizing hearts from DCD donors, hearts that were previously discarded because they were determined to be too injured and too high risk for subsequent problems. Warm blood perfusion technologies have made it possible to recover DCD hearts and support them for four hours prior to transplantation. This allows Vanderbilt thoracic organ recovery teams to travel farther in search of organs and add hundreds of organs to the donor pool. About half of Vanderbilt’s heart transplants in 2023 used DCD organs.

“The Vanderbilt Transplant Center is committed to providing life-saving organ transplants to as many needy patients as possible,” said Joseph Magliocca, MD, professor of Surgery and director of the Vanderbilt Transplant Center. “Unfortunately, transplantation has long been limited by a large discrepancy between the number of organs available and the number of patients waiting. One of our most active areas of research has been exploring ways to improve the quality of donor organs. These technologies have allowed us to transplant organs that would have previously been discarded. Our physicians and surgeons are truly on the cutting edge of the development of novel organ preservation methods.”
Unlike Vanderbilt’s previous DCD heart transplants, which have used a warm blood perfusion system, the XHAT technology perfuses hearts at a colder temperature with a specialized preservation solution. This may have important advantages over conventional storage on ice or the warm system, Shah said.
“Perhaps the most important advantage of this is that it’s far simpler to manage than any than other technology that’s out there,” he said. “Warm perfusion is an excellent technology, but without the protection afforded by hypothermia, it makes the heart vulnerable to injury. So, everything has to be perfect. In a cold, perfused heart, you have another layer of protection.”
Shah said the technology may also allow donor hearts to be preserved even longer. Conventional cold and static storage allows for safe preservation under four hours. This novel cold perfusion platform, however, has already allowed Vanderbilt to protect hearts past seven hours. More time allows the medical team to find the best match for donor organs and to study techniques to better preserve and perhaps repair organs during perfusion.
“For decades, time has been our enemy when it comes to organ donation,” said Christopher Schwartz, RN, organ recovery coordinator and lead organ perfusionist for the heart, lung and liver transplant programs. “With this new technology along with the multidisciplinary team we have put together at Vanderbilt, this is no longer the case. There is literally no limit to where we can go in the U.S. now to recover hearts for our patients.”
Prior to 2020, Vanderbilt only transplanted organs from DBD (donation after brain death) donors. Like DCD donors, DBD donors have sustained devastating, non-recoverable neurologic injury. Unlike DBD donors, however, DCD donors don’t yet meet formal brain death criteria — as such, the methods that are used for withdrawal of donor life support and surgical retrieval of DCD versus DBD organs differs.
Until recently, there had been limited data on clinical outcomes in recipients of DCD as compared to DBD heart transplants. But a VUMC study published in October 2023 in the Journal of the American College of Cardiology found no difference in one-year survival and other outcomes among DBD and DCD donors.
“Part of our mission is not only to transplant more people, but to actively improve heart transplantation,” Shah said. “This study, and particularly this case, highlights how much we still don’t know about organ transplantation and how to make it better, safer, more durable and more widely applicable.”
All this is made possible by Vanderbilt’s thoracic transplant recovery team. This group has matured into a multidisciplinary clinical powerhouse that includes preservationists, surgeons and perfusionists.
“These are extremely exciting times to be in the field of solid organ transplantation,” said Matthew Warhoover, CCP, chief of Perfusion. “There have been tremendous strides over the last few years, but what I find fascinating is even with this, we continue to ask, ‘Can we do something better for the donor organ?’ It’s this thought process that can translate in providing the organ recipient with a better outcome.”
VUMC’s Stephen DeVries, DMSc, PA-C, a physician assistant who works in organ recovery, added, “Since 2019, the field of donor organ recovery and preservation has exploded and continues to evolve rapidly. As a worldwide leader in heart transplantation, it is our responsibility to help determine how to best apply and utilize these new technologies not only for our own program, but for the field of heart transplantation as a whole. Dynamic cold storage with XVIVO is the latest new technology to be introduced in the United States currently under clinical trial.”