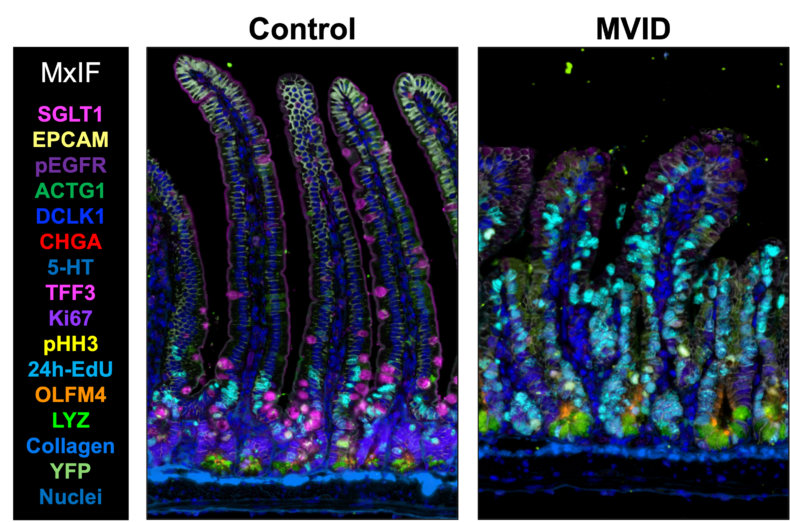
 Multiplex immunofluorescence (MxIF) images of the small intestine of healthy (control) and MYO5B-deficient (MVID) mice. MVID patient biopsies and mouse models demonstrate the expansion of proliferative zones and decreases in nutrient sensory and absorptive cells, resulting in diarrhea symptoms. (image courtesy of Izumi Kaji, PhD)
Multiplex immunofluorescence (MxIF) images of the small intestine of healthy (control) and MYO5B-deficient (MVID) mice. MVID patient biopsies and mouse models demonstrate the expansion of proliferative zones and decreases in nutrient sensory and absorptive cells, resulting in diarrhea symptoms. (image courtesy of Izumi Kaji, PhD)
Microvillus inclusion disease (MVID) — a rare genetic disorder characterized by severe, watery diarrhea and the inability to absorb nutrients — is caused by mutations in the motor protein myosin VB (MYO5B). MVID is fatal without treatment, which is currently limited to lifelong parenteral nutrition (IV delivery of essential nutrients) or intestinal transplantation.
Izumi Kaji, PhD, assistant professor of Surgery, and colleagues are studying the function of MYO5B in the intestine, with the goal of identifying alternative, less invasive treatment options for MVID.
Previous studies have demonstrated the importance of MYO5B in the development of the intestinal brush border — the nutrient-absorbing surface of intestinal epithelial cells. But the protein’s role in intestinal crypts, where proliferative progenitor (stem) cells give rise to the mature cell types of the small intestine, has remained largely unexplored.
Kaji and colleagues have now developed special mouse models that lack MYO5B in intestinal crypt cells. Using transcriptomic approaches and multiplex immunofluorescence, they demonstrated that MYO5B is critical for intestinal stem cell identity and function.
They showed that crypts missing MYO5B have excessive cell division and incomplete cell differentiation in vivo compared to control crypts and that they form organoids in vitro at an elevated rate. MVID patient biopsies also showed abnormal expansion of the crypt proliferative zone. The researchers reported that disruptions in mitochondrial structure and fatty acid metabolism likely underlie crypt dysfunction and that deficits are distinct from starvation.

The findings, published in Stem Cell Reports and featured on the journal cover, suggest that future MVID therapeutics should focus on promoting proper intestinal stem cell function to correct disease-related disruptions in small intestinal epithelial architecture, the researchers note.
Andreanna Burman, PhD, from the Department of Cell and Developmental Biology at Vanderbilt University and the Epithelial Biology Center at Vanderbilt Health, is the study’s first author; Kaji is the senior and corresponding author. Co-authors are Monica Brown, Yilin Yang, Michael Momoh, Francisca Adeniran, Cynthia Ramos, Ken Lau, PhD, and Joseph Roland, PhD, from Vanderbilt University and Vanderbilt Health; Linda Samuelson, PhD, from the University of Michigan; and Mitchell Shub, MD, from the University of Arizona College of Medicine and Phoenix Children’s.
The research was supported by the National Institutes of Health (grants R01DK128190, RC2DK118640 and R01DK103831) and the National Science Foundation.






